A configuration discussed here entails the daily operation of a small wood gas engine system for 3-5 hours, or however long the requirements and/or fuel availability permits.
First, let's assume that air conditioning is desired. I propose that a wood gas engine system be used to drive an alternator (for battery charging), but with most of the engine power used for driving an automotive a/c compressor for partially freezing a mass of water kept in an insulated tank. This cold source can be tapped for a mini chilled water system. BTW, these automotive compressors are inexpensive and durable. They can be used to make an a/c system with impressive performance by using large heat exchangers and cooling them well. Automotive a/c systems do not have high performance because the heat exchangers have to be very compact and they are not well cooled behind the fire wall of an engine compartment. Furthermore, this a/c system can be operated at night when air temperatures are lower for good condenser cooling. Also, it makes sense to do this at night since battery charging can be done as well, and this will minimize battery discharge since a modest solar array can be putting out during the day. Finally, since the heat from the engine system is not put to much use here, then a good way to use it is for fuel drying, water processing (i.e. pasteurization) and/or heating, and some heat might be used to regenerate a desiccant to help lessen the latent cooling load in humid climates.
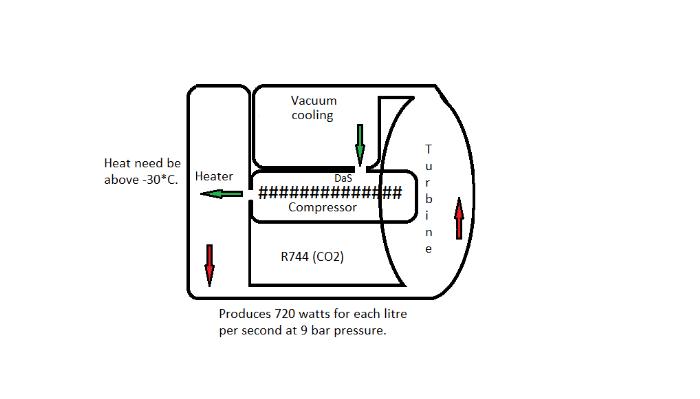
Now, about heating. A heat exchange system can be devised to catch most of the heat from a wood gas engine system. A good example is Ken Boak's system (google Ken Boak, lister, powercubes). Water is pumped to catch the heat from the cylinders, wood gasifier, and engine exhaust and this heated water/steam can be used to heat the store of water that was used for cooling, then use a hydronic air heating system (i.e. using the chilled water system with hot water flowing through the coils instead of cold). However, since the output from the engine has to be fairly high to ensure enough air is pulled through the gasifier at all times, then this additional energy must be put to use (note: generally, a small wood gas engine system needs to operate at a rate of at least 5 hp to prevent having to use a very small gasifier that might require pellets or other highly processed particulate biomass). There is no a/c compressor here, and only very large battery systems can handle this kind of charge rate (would be on the order of 2500-3000 watts). So, the shaft energy normally applied to the a/c compressor could be used to generate more electricity for directly heating with electricity, and this would provide heat at a rate of about 1500-2000 watts, OR one can put a cheap modified sine wave inverter on the battery to power conventional space heaters to draw down the battery during charging and reduce the net charge rate, OR it's also possible to configure the a/c system for heating by placing a condenser in the home, and this would provide a lot more heat by transferring heat from outside to within this home. However, this latter option is not the best where outside temperature are particularly low. The idea here is that a condenser OR electric heat could provide a rapid heat up of a home while the engine system is operated each day, then the remaining heat from the engine could be stored up to provide heat over the rest of the 24 hour period.
In other words, a wood gas engine system can be configured to work exceptionally well in the off grid residential setting, but I don't recommend trying to adapt such a system towards powering a modern home in a conventional manner (i.e. primarily with electricity). One must put the heat to use where possible/practical. It would be wonderful if a wood gas engine system can be operated continually at low power, but this is really a very difficult if not impractical goal. Rather than force a square peg into a round hole, perhaps it's best to adapt a configuration that optimizes existing wood gas engine systems.
NOTE: On water processing with pasteurization, I was considering that passing the heated water (heated from the wood gas engine system) through a small thermal mass before it moves to other heating applications. Once the thermal mass reaches a preset temperature, then a thermostat can shut to energize the motor of a water pump and start sending water through this heated mass. Providing this thermal mass can simplify the water pasteurization process. BTW, I'm considering a setting where the home is situated near a body of fresh water that can be drawn upon as required. Pasteurization might also be useful for processing water from shallow wells. It might even be used as a preventive measure for processing all off grid water sources. After all, since one desires to heat the water anyway, then might as well jack up the temperature for a while to kill any bugs that might be lurking. Use heat regeneration to reduce energy requirements here, but the water can retain enough heat when stored in an insulated tank so that it would not require additional heating after the pasteurization process (instead of on demand heating of cool water when hot water is desired, try on demand cooling of hot water when cool water is desired... it's easier to cool water than to heat it,

. So, store the water while it's still pretty hot. NOTE: Heat regeneration here entails the preheating of the incoming water with the water that is pasteurized to reduce the overall heat required to process a given mass of water. So, take the water to high temp with the thermal mass, then cool it down some by transferring some heat to the cool water before it moves into the heater.
NOTE: On desiccant regeneration, getting the heated water (used to harvest heat from the wood gas engine system) to the highest possible temperature, then passing this water through the desiccant solution FIRST (even before the thermal mass used for pasteurization) could be done. This will moderate the temperature somewhat before the hot water moves to the other applications. Also, since this is desired only when a/c is desired (i.e. summer months), then getting as much heat as possible into the desiccant should be done during these times. A desiccant can be very useful to help remove some moisture from the air in a humid environment and enhance the cooling effect of the a/c system.





 . So, store the water while it's still pretty hot. NOTE: Heat regeneration here entails the preheating of the incoming water with the water that is pasteurized to reduce the overall heat required to process a given mass of water. So, take the water to high temp with the thermal mass, then cool it down some by transferring some heat to the cool water before it moves into the heater.
. So, store the water while it's still pretty hot. NOTE: Heat regeneration here entails the preheating of the incoming water with the water that is pasteurized to reduce the overall heat required to process a given mass of water. So, take the water to high temp with the thermal mass, then cool it down some by transferring some heat to the cool water before it moves into the heater.
 1
1